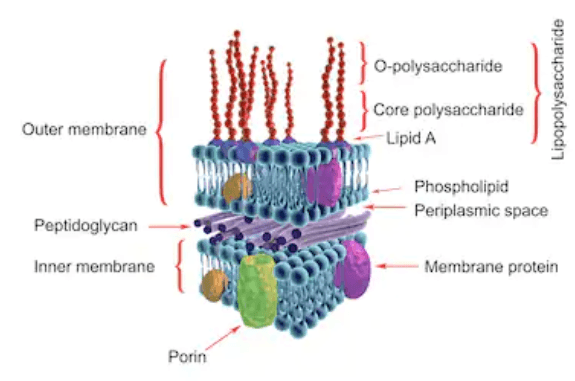
Endotoxins are toxins present within a bacterial cell wall that are released when a cell disintegrates. They are lipopolysaccharides (LPS) and are found within the outer membrane of the cell wall of Gram-negative bacteria. Endotoxins often consist of lipid A, a core oligosaccharide and the O-specific polysaccharide.
Why Endotoxins are a Problem
When the outer membrane of the bacterial cell disintegrates, endotoxin is released into the bloodstream which in turn releases cytokines. All three components of the LPS molecule are immunogenic, provoking an immune response by eliciting the formation of antibodies that interact with the binding site of antigen molecules, known as the epitope. When LPS-sensitive cells interact with endotoxins, this can result in fever, inflammation, sepsis, tissue damage and in serious cases, death. Endotoxins can form stable interactions with other biomolecules, making their removal extremely difficult, particularly during the production of medical devices and pharmaceutical drugs.
Detecting Endotoxins
Endotoxins can be detected using the Limulus Amoebocyte Lysate (LAL) test. LAL exists in the blood of horseshoe crabs and forms a blood clot when exposed to Gram-negative pathogens. The LAL test employs a protease cascade initiated by the combination of LPS and zymogen Factor C. The pro-clotting enzyme is converted to the clotting enzyme and two peptide bonds in coagulation are catalytically cleaved to form coagulin gel. The U.S. Food and Drug Administration (FDA) permit the LAL test, known as Bacterial Endotoxin Testing (BET), to be used to detect endotoxin in finished pharmaceuticals, raw materials and medical devices. Medical devices that come into direct or indirect contact with blood are safety tested by testing for endotoxins via an extraction process used to flush away material ready for lysate reagent binding (<USP 161>).
Advanced LAL techniques are also available, including an animal-free, FDA-approved method:
- Kinetic Turbidimetric LAL assay
- Endpoint Chromogenic LAL assay
- Kinetic Chromogenic LAL assay
- Recombinant Factor C assay (animal-free, FDA-approved)

Below are the standards references by the FDA and MHRA (Medicines and Healthcare products Regulatory Agency) for bacterial endotoxin testing:
- USP 85: Bacterial Endotoxin Test: gel-clot techniques and photometric techniques are described.
- USP 161: Transfusion and Infusion Assemblies – outlines limits, provides definitions and references test methods required.
- AAMI ST 72:2011: Bacterial endotoxins – Test methods, routine monitoring and alternatives to batch testing.

When to use Endotoxin Testing
Endotoxin testing is used during medical device manufacturing, pharmaceutical manufacturing, packaging manufacturing and raw materials production. US and European regulations state that to test medical devices for endotoxin, it will have to be extracted/rinsed from the surface of the device. The effects of endotoxin relate to the amount of endotoxin within the product dose, which varies from product to product. Endotoxin limits are therefore used to outline the dosage threshold for humans.
If you would like to know more about Perfectus Biomed’s Endotoxin Testing services, please get in touch.